
Hydrogen is a fantastic energy carrier. It is a clean fuel: during combustion, the only waste product is pure water. There is no CO₂ released into the atmosphere. Theoretically, there is plenty of hydrogen available. You can make it in several ways, even from water using electricity. That electricity can now be produced from renewable sources like wind and solar power. In a hydrogen fuel cycle, water is split into hydrogen and oxygen by the process of electrolysis and then they re-combine, producing electricity and water vapour. So, why hydrogen is not widely used in our energy ecosystem?

Wind power plants can produce green hydrogen
By using hydrogen, we can prevent uneven energy supply from certain renewable sources. When the sun is shining and wind is blowing, we can produce hydrogen with surplus energy, and use it when there is demand but no direct solar or wind power available.
We can even transfer energy from one place to another in the form of hydrogen. In the future, this may help us to avoid heavy investments in new power grids from windy coastal areas to industrial regions elsewhere.
Hydrogen power will come. Japan and South Korea have already established roadmaps to incorporate hydrogen as a major fuel in their economies, creating immediate demand for clean hydrogen in bulk. The price of key technology components is coming down. Earlier this month, ministers of the G20 countries promised to step up existing international efforts to utilise hydrogen. [1]
However, so far we have not used hydrogen very much as a fuel. Let’s take hydrogen cars as an example: since the 1960's, when General Motors introduced its first fuel-cell vehicle concept, mass-market hydrogen cars have always been said to be about ten years away. Today only very few cars using hydrogen roll on our streets. Why is this?
Despite its great potential, hydrogen is a somewhat complicated fuel to use. There are applications in which it already makes sense to use hydrogen and then there are others that still need further technological development. Hydrogen cars fall to the latter category.
Hydrogen burns fast
You have to be very careful when handling hydrogen, as was proven this month when a hydrogen station exploded in a suburb of Norway's capital, Oslo. [2]
Hydrogen explosions can be easily triggered because of the high flammability of hydrogen. This is partly caused by the very wide flammability range (the portion of fuel mixed in air to form a flammable concentration) of hydrogen compared to other fuels. Hydrogen also requires only approximately one-tenth of the energy needed to initiate combustion compared to other standard fuels. [3]
And when there are explosions, they can be severe: hydrogen combustion is much more rapid than that of other fuels – approximately ten times quicker.
In energy production, flame speed causes many challenges. For many technologies, hydrogen just burns too fast and therefore the use of it is either impossible or the full benefit of the fuel cannot be reached.
Have a look at the graph below to see the differences between the burning velocities of methane (CH4), hydrogen (H2) and carbon monoxide (CO). ‘Air excess ratio’ defines the amount of air used by the fuel; when this ratio is 1.0, all the oxygen in the air is being used in combustion. Please note the scale is logarithmic. The graph shows clearly that the combustion of hydrogen is almost explosion-like compared to other fuels.
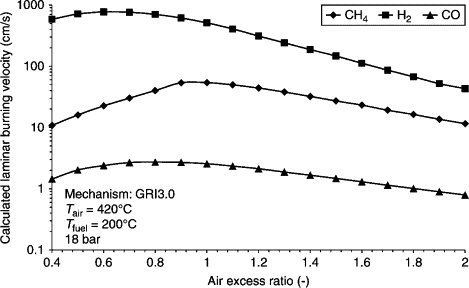
In reciprocating engines, you must compensate for this somehow. You can do this, for instance, by mixing hydrogen into another fuel or by enhancing the air excess ratio by using less fuel in a larger amount of air (over-stoichiometric combustion). This compensation will either lessen the benefits of the fuel or generate less power from the engine.
A good example of this is BMW Hydrogen 7. It was a limited production hydrogen internal-combustion engine vehicle built from 2005-2007 by BMW. The car was powered by a 6.0-liter V12 engine, but it only reached 256 horsepower. [5]
It requires a lot of energy to compress hydrogen
Hydrogen can be stored and used as a liquid or slush as in the Space Shuttle. However, either requires extremely low temperatures because hydrogen boils at approx. −253 °C. [6]
It is more common to store it as a compressed gas in high-pressure tanks. This presents its own challenges: for instance, you’ll need a relatively large tank for storage. In many hydrogen cars, the tank is surprisingly large and inconveniently shaped, restricting luggage space.
Even though hydrogen has good energy density by weight, it has poor energy density by volume versus hydrocarbons, even gaseous ones. As an example, at any pressure, the volumetric energy density of hydrogen is 3.2 times less than that of methane.
While the behaviour of most gases can be predicted using the ideal gas equation (PV = nRT), hydrogen’s behaviour differs significantly. This deviation is the result of expansion – hydrogen demands more space than the equation predicts. The graph below shows the necessary compression energy of hydrogen versus helium and methane. [7]
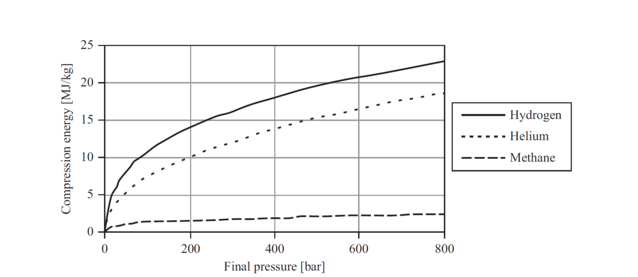
Because hydrogen compression requires energy and the volumetric energy density requires high pressures, there are inevitably high losses in hydrogen compression; a developed multi-stage compression to 500 bar will consume approx. 10% of the higher heating value of the fuel. [8]
Hydrogen can come through the walls
Hydrogen is the first, smallest and lightest of all the elements. This means that hydrogen can enter and diffuse through steel even at room temperature [9]. Hydrogen can also penetrate right into the crystal structure of a solid metal [10], which can embrittle steel and will cause the rigidity of the steel to weaken [11]. This combined with the requirement to withstand high pressures makes many ordinary steel structures unfit for storing hydrogen. Luckily new materials and methods have been developed to overcome this issue.

Turbine technology can solve several issues related to the use of Hydrogen
Several problems related to the use of hydrogen have lately been partially or completely solved. One problem still unsolved is how to use hydrogen efficiently in decentralised power generation.
Because of the reasons described above, hydrogen cannot be utilised efficiently in reciprocating engines. Additionally, the traditional combustion chambers and fuel nozzles used in gas turbines are not sufficient. For example, if pure hydrogen was used with typical natural gas fuel nozzles in a turbine, the nozzles would melt as the combustion would take place far too close to the nozzles. Rather than burning in the combustion zone, the hydrogen would burn in the nozzle itself.
Unlike reciprocating engines, gas turbines have one big advantage: their combustion process is not intermittent, but stable and on-going. Thus, completely new approaches can be utilised to fine-tune the turbine process to accommodate the fuel. One possible solution utilises flameless combustion chambers, as shown below.

On top of being a completely CO₂ free fuel, the use of turbine technology will also minimise the nitrogen oxide emissions.
-----------------------------------------------------------------------
- https://www.pv-magazine.com/2019/06/10/green-hydrogen-airs-at-g20-meeting-of-energy-ministers-this-week/
- https://www.autoblog.com/2019/06/12/norway-hydrogen-station-explodes-toyota-hyundai-halt-sales/
- https://h2tools.org/bestpractices/hydrogen-compared-other-fuels
- https://www.sciencedirect.com/topics/engineering/flame-speed
- https://en.wikipedia.org/wiki/BMW_Hydrogen_7
- https://en.wikipedia.org/wiki/Hydrogen_storage
- https://www.researchgate.net/profile/Sofoklis_Makridis/publication/313704619_Hydrogen_storage_and_compression/links/5a7b2ac1458515229b1c303d/Hydrogen-storage-and-compression.pdf
- https://afdc.energy.gov/files/pdfs/hyd_economy_bossel_eliasson.pdf
- https://www.imetllc.com/training-article/hydrogen-embrittlement-steel/
- https://phys.org/news/2019-02-hydrogen-effects-metal.html
- https://www.mig-welding.co.uk/hydrogen-embrittlement.htm
